 3 - 6īecause of the safety and efficacy data of clinical trials for ES-SCLC, chemoimmunotherapy has been used for patients in clinical practice. 3 Consequently, chemoimmunotherapy, ie, the addition of PD-L1 inhibitors to the combination of a platinum agent and etoposide, has become a standard first-line treatment option for ES-SCLC. 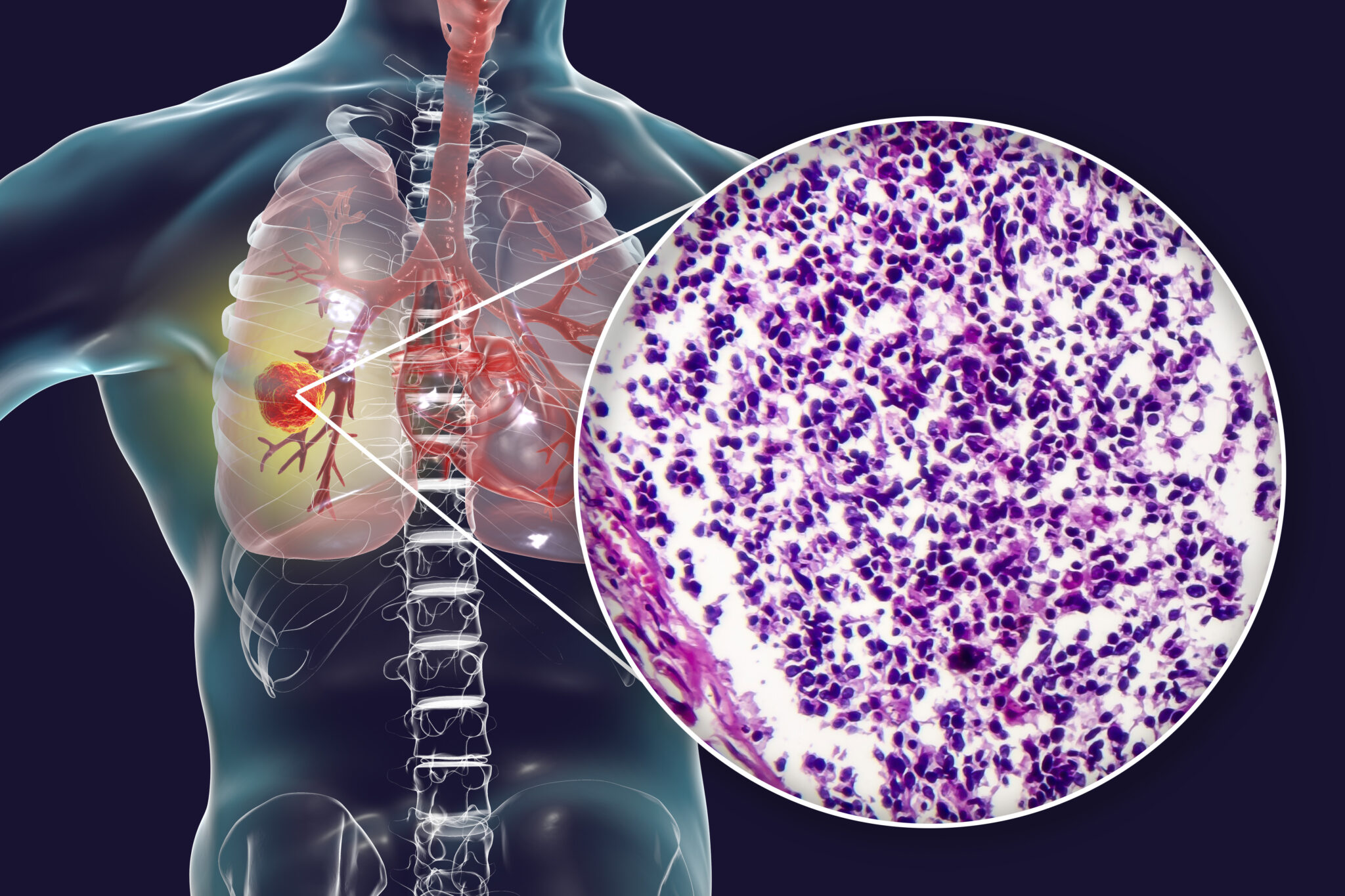
In a pivotal phase 3 trial, the addition of atezolizumab to chemotherapy resulted in longer progression-free survival (PFS) (5.2 vs 4.3 months P = .02 hazard ratio, 0.77 95% CI, 0.62-0.96) and overall survival (12.3 vs 10.3 months P = .007 HR, 0.70 95% CI, 0.54-0.91) than chemotherapy. 1, 2 The development of programmed cell death protein 1 (PD-1)/programmed death-ligand 1 (PD-L1) checkpoint inhibitors has markedly changed the treatment strategy for ES-SCLC. 1 Small cell lung cancer (SCLC) accounts for 15% to 20% of all lung cancer cases, and extensive-stage SCLC (ES-SCLC) accounts for approximately two-thirds of all SCLC cases. Lung cancer is the leading cause of cancer-related deaths worldwide. These findings suggest that trial-eligibility criteria may be useful in clinical practice, and further studies using data from clinical practice settings are required to inform regulatory approval and clinical decision-making. However, treatment outcomes after chemoimmunotherapy might differ between trial-eligible and trial-ineligible patients. The rate of severe adverse events was numerically higher among trial-ineligible patients than among trial-eligible patients (39% vs 27% P = .07).Ĭonclusions and Relevance In this cohort study, the overall treatment outcome was comparable to that reported in pivotal clinical trials. The median overall survival was 15.8 months in trial-eligible patients and 13.1 months in trial-ineligible patients (hazard ratio, 0.73 95% CI, 0.51-1.07 P = .10). 
The proportion of patients who achieved disease control was 93% (118 of 127) in trial-eligible patients and 77% (55 of 71) in trial-ineligible patients ( P = .002). The median progression-free survival was 5.1 months in trial-eligible patients and 4.7 months in trial-ineligible patients (hazard ratio, 0.72 95% CI, 0.53-0.97 P = .03). The 6-month progression-free survival rate for all patients was 38.8% (95% CI, 32.4%-45.7%). There were 132 (64%) trial-eligible patients. Sixty-four patients (31%) were older adults (age ≥75 years), and most (184 ) had an Eastern Cooperative Oncology Group performance status of 0 or 1. Results A total of 207 patients were analyzed (median age, 72 years range, 46-87 years 170 were male). The secondary outcomes were differences in progression-free survival, overall survival, and safety according to whether key clinical trial eligibility criteria were met. Main Outcomes and Measures The primary outcome was 6-month progression-free survival. Participants included consecutive patients with ES-SCLC who received carboplatin and etoposide with atezolizumab as first-line therapy.Įxposures Patients who met eligibility criteria for pivotal phase 3 clinical trials were considered trial-eligible. Objective To compare treatment outcome gaps following first-line chemoimmunotherapy for patients with ES-SCLC between those who met and did not meet the eligibility criteria used in previous clinical trials.ĭesign, Setting, and Participants A prospective cohort study was conducted from September 1, 2019, to September 30, 2020, at 32 hospitals in Japan, with at least 12 months of follow-up. However, whether findings from pivotal trials can be extrapolated to the clinical practice setting remains unclear. Importance Chemoimmunotherapy is the standard first-line therapy for patients with extensive-stage small cell lung cancer (ES-SCLC). Shared Decision Making and Communication.Scientific Discovery and the Future of Medicine.Health Care Economics, Insurance, Payment.Clinical Implications of Basic Neuroscience. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
